Elon Musk’s Neuralink company seems to be advancing closer to testing its brain-computer interface technology in humans with the posting of a new job listing. Neuralink is now recruiting for a clinical trial director who will “work closely with some of the most innovative doctors and top engineers, as well as working with Neuralink’s first Clinical Trial participants!”
About Neuralink
Neuralink is a team of exceptionally talented people. We are creating the future of brain-machine interfaces: building devices now that will help people with paralysis and inventing new technologies that will expand our abilities, our community, and our world.
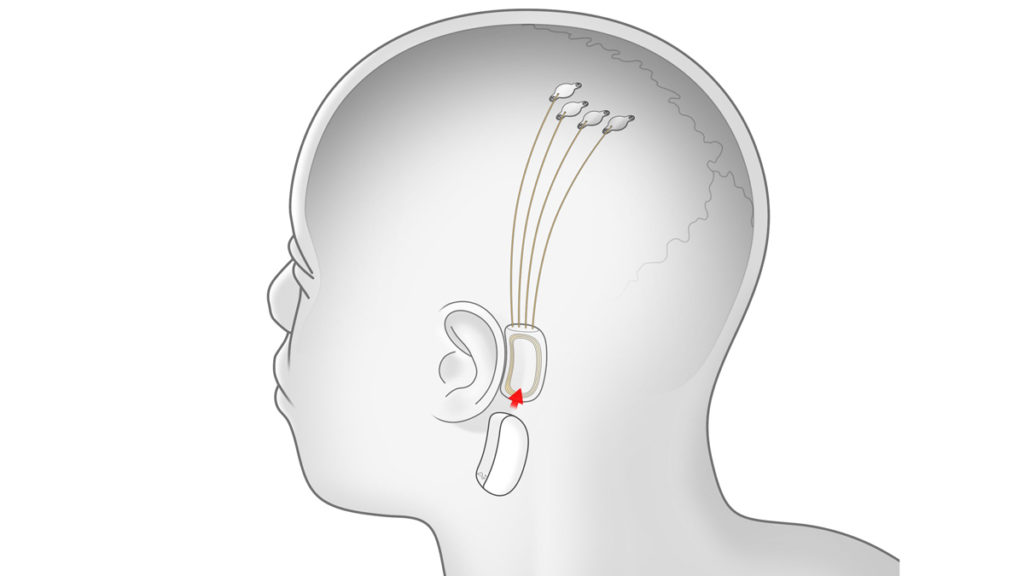
Our goal is to build a system with at least two orders of magnitude more communication channels (electrodes) than current clinically-approved devices. This system needs to be safe, it must have fully wireless communication through the skin, and it has to be ready for patients to take home and use on their own. Our device, called the Link, will be able to record from 1024 electrodes and is designed to meet these criteria.
Neuralink recently had demonstrated its technology being used on a monkey who had managed to play Pong via the implant. Mr. Musk attended the Wall Street Journal CEO Council Summit and said to attendees, “[w]e hope to have this in our first humans, which will be people that have severe spinal cord injuries like tetraplegics, quadriplegics, next year, pending FDA approval.” He added that currently it is “working well in monkeys, and we’re actually doing just a lot of testing and just confirming that it’s very safe and reliable and the Neuralink device can be removed safely.”
Elon has gone on record to explain that the implant should be unnoticeable in humans and that it would be “flush with skull & charges wirelessly, so you look and feel totally normal.” He hopes that it could potentially help those who’ve experienced a brain or spinal injury regain some level of mobility. Some people have already asked if it may even help with brain disorders, to which he replied that “[r]eplacing faulty/missing neurons with circuits is the right way to think about it” and that “[m]any problems can be solved just bridging signals between existing neurons.”
Replacing faulty/missing neurons with circuits is the right way to think about it. Many problems can be solved just bridging signals between existing neurons.
— Elon Musk (@elonmusk) December 7, 2021
Progress will accelerate when we have devices in humans (hard to have nuanced conversations with monkeys) next year.
However, getting FDA approval for testing may only be one of many hurdles in getting human clinical trials rolling. A quick glance at all the job listings for Neuralink shows the company has a sizable amount of positions to fill.


Source: Neuralink (1, 2) (via The Guardian)
